
Watch our Webinar: The power of nutrition and mitochondrial diseases
3 Oct, 2025
Watch our Webinar: The power of nutrition and mitochondrial diseases As part of LHON Awareness Day, we hosted a powerful […]...

From Advocacy to Access: Mito Community Celebrates FDA Milestone
26 Sep, 2025
Barth syndrome therapy sets precedent for future treatments targeting mitochondrial dysfunction In a landmark decision, the U.S. Food and Drug […]...
IMP Launches New TK2d Video
11 Sep, 2025
IMP Launches New TK2d Video To mark TK2d Awareness Day 2025, we’ve released a new video that amplifyies the voices […]...
IMP Welcomes Three New Members
9 Sep, 2025
IMP now has members across three continents We’re delighted to welcome three new members to IMP. With these new members, […]...

Landmark Success for Mitochondrial Donation: A Milestone for Families Worldwide
17 Jul, 2025
The birth of eight healthy babies in the UK through mitochondrial donation (also known as Mitochondrial Replacement Therapy) has been […]...

IMP Announces New Board Following Election
15 Jul, 2025
IMP is delighted to announce the results of its recent Board elections, confirming a dynamic and diverse leadership team that […]...

19 May, 2025
Scientists provide first evidence that the drug sildenafil may benefit patients with Leigh syndrome, and particularly those carrying mutations in the mitochondrial gene MT-ATP6....

Primary Mitochondrial Myopathies: Patient Journey
20 Dec, 2024
Navigating the journey to gain a diagnosis of Primary Mitochondrial Myopathy can be unnecessarily long, intensely stressful, and fraught with obstacles....

Countdown to Rare Disease Day 2025!
13 Dec, 2024
Collaboration with MITGEST
9 Dec, 2024
IMP is proud to support MITGEST. This international consortium brings together world-class academic experts in mitochondrial research....

SIMPATHIC project prepares to launch clinical trial
9 Dec, 2024
The SIMPATHIC project is preparing to launch a clinical trial using Sildenafil as a potential treatment for Leigh syndrome. ...

IMP joins UK MitoCohort Oversight Committee
9 Dec, 2024
IMP are delighted to have been asked to join the UK MitoCohort Oversight Committee. The UK Mitocohort is a UK wide database that collects clinical information on patients living with mito, to help advance understanding of the disease....

The PolG Foundation: Advancing Research and Hope for POLG Disorders
27 Nov, 2024
The PolG Foundation is committed to accelerating research efforts to develop effective treatments and, ultimately, a cure for POLG mitochondrial disorders....

LHON Awareness Day Webinar
18 Nov, 2024
During this year’s World Mitochondrial Disease Week, IMP hosted a free webinar on 19 September for LHON Awareness Day. The theme for the webinar was LHON and Gene Therapy: Where are they now? ...

Khondrion announces publication of results of integrated Phase 2b programme
15 Nov, 2024
On 7 November, biopharmaceutical company Khondrion announced the publication of results from their integrated Phase 2b clinical development programme which uses the drug sonlicromanol to target the underlying pathways of primary mitochondrial disease....

World Mitochondrial Disease Week 2024
30 Oct, 2024
We had an amazing World Mitochondrial Disease Week this year. This year’s Awareness Week theme was Illuminate Tomorrow: Revitalize your energy. From 16-22 September, our member organisations and community around the world hosted events and took to social media to raise awareness about mito....

Updated TK2d infographics to download
9 Aug, 2024
Help raise awareness of the symptoms of TK2D by sharing our fantastic infographics. We've slightly updated these from 2023, so please do replace your older graphics with these ones. Download the version in English or landscape version in English, or follow the links for versions in Italian, Spanish, and German:...
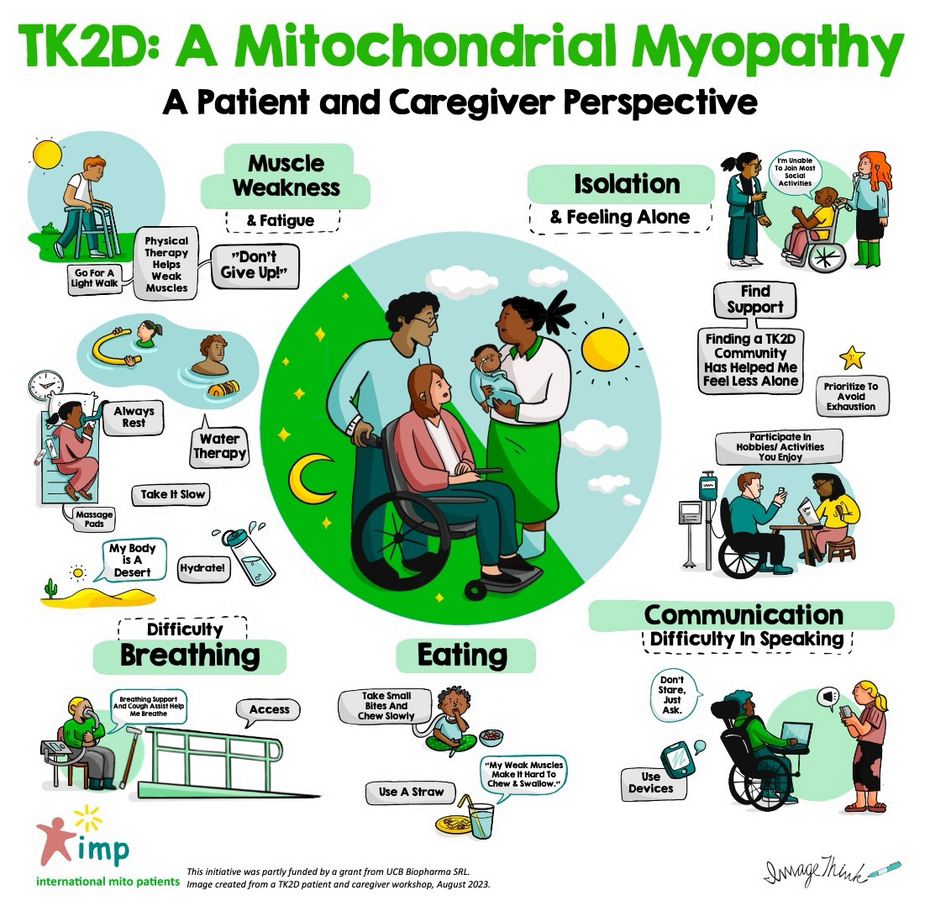
Seeking the patient perspective: Living with LHON
9 Aug, 2024
Hello there The results of the forums have been published in two reports (see links below). They captured the key discussions and provided people living with LHON the important opportunity to connect on critical issues, share experiences and make recommendations....

LHON webinar – Save the date!
9 Aug, 2024
Theme: LHON & Gene Therapy: Where are we now? Save the date for a free, interactive webinar discussing gene therapy and LHON. Hear the latest from companies developing gene therapies for LHON, clinical experts and someone who has been part of a gene therapy clinical trial. And find out what you can do to ensure the voice of people living with LHON is heard! Our webinar will be on 19 September 2024 from 13:00 - 15:00 CEST. To register your interest click on the link below....

Italy leads way for legislation re mito DNA replacement
27 Feb, 2024
News from Mitocon: Italy leads the way in the European Union with a secondary legislation for testing mitochondrial DNA replacement techniques...
